ntThe work function of metal is 1 eV. Light of wavelength 3000 is incident on this metal surface. The velocity of emitted photo electrons will ben

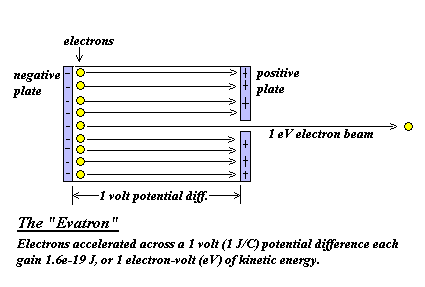
Electron volt is the unit of energy (1 eV=1.6times 10^{-19}J). In H-atom, the binding energy of electron in first orbit is 13.6 eV. The same in joule (J) is?